Coles Miller Solicitors crowned champions at Poole’s Bowls Bash 2025
Poole, Dorset, August 2025



£30,000 settlement secured for pedestrian injured in e-scooter collision

Bringing dreams to life: Coles Miller facilitated a dream come true for Charlie, who suffered from cerebral palsy due to a birth...

Coles Miller support Forest Holmes Hospice

January 2025 – Forest Holme Hospice Charity’s second Strictly Extravaganza dazzled audiences at the Poole Lighthouse on Saturday...

Remember lost loved ones at this Friday 8 November 2024 (7pm) candlelit event in aid of University Hospitals Dorset NHS Charity....

Teams from across Dorset and the New Forest have raised vital funds for Forest Holme Hospice at Coles Miller's Charity Quiz Night....
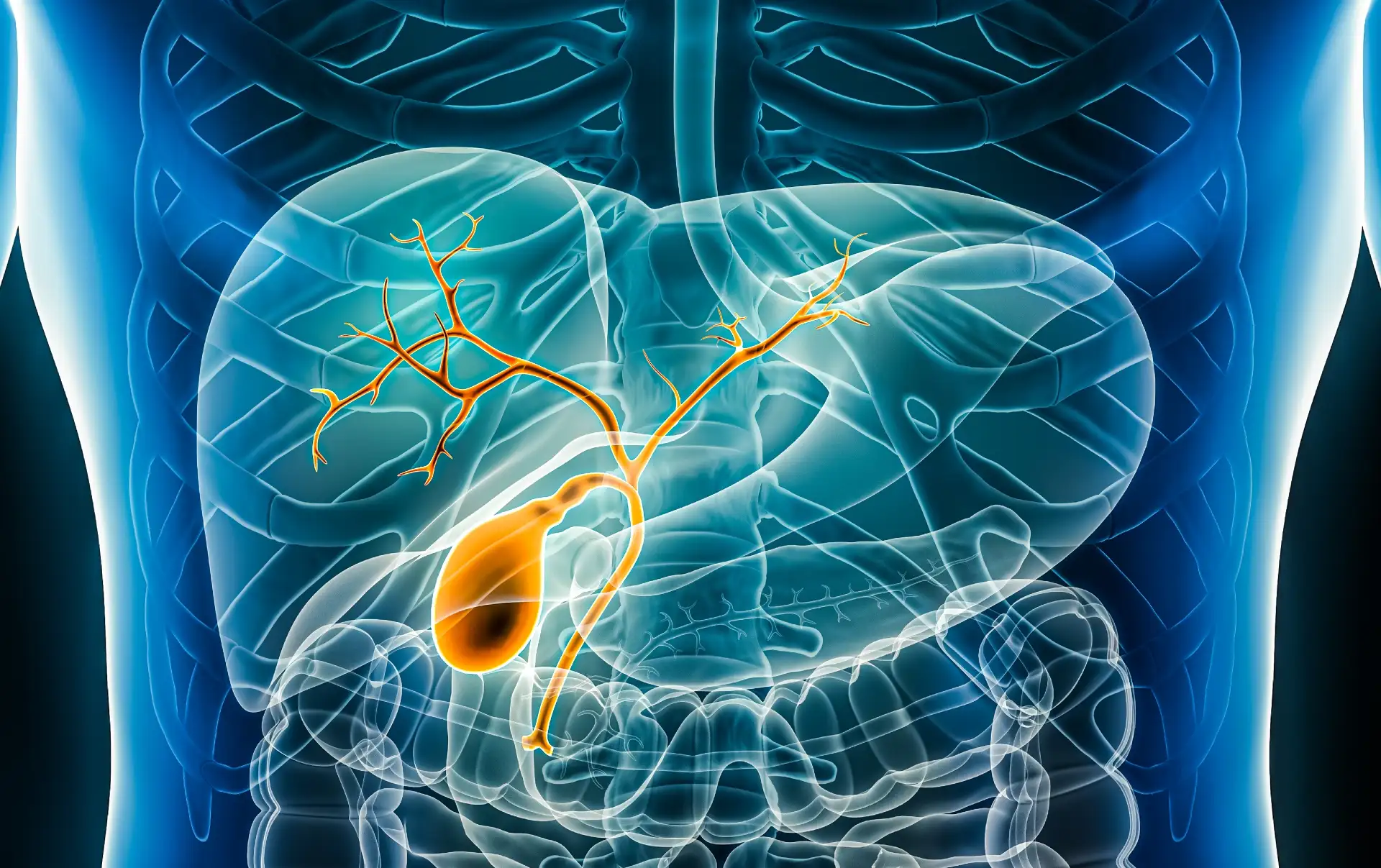
Coles Miller medical negligence solicitors helped a grandmother to claim £100,000+ compensation after her gallbladder operation...
.png)
September 19, 2024

Dorset law firm Coles Miller has been named as the first Hidden Disability Friendly business by charity Growing Compassionate...

Record numbers of dementia cases make it more important than ever to have valid powers of attorney and an up-to-date will, warn...